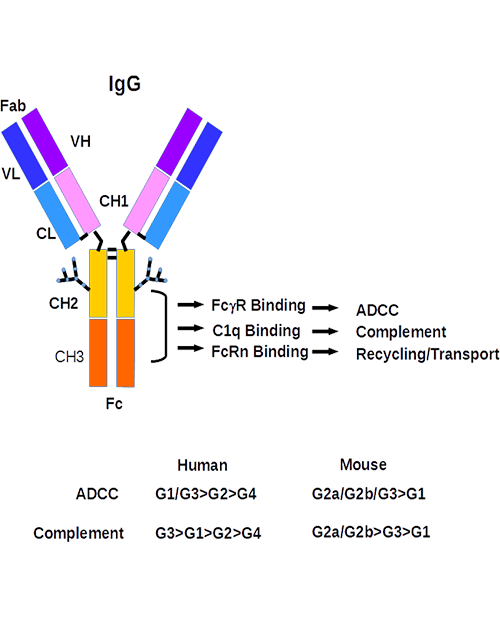
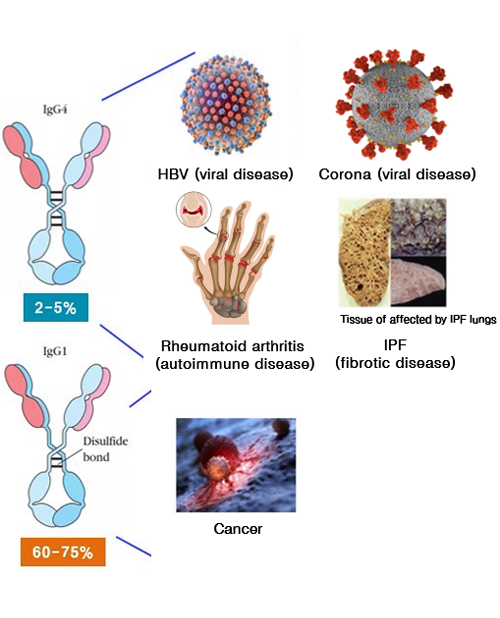
Antivirals
The current antiviral market is about 30 trillion KRW (23 billion USD). A significant portion of viral diseases are intractable diseases, and the growth potential of the antiviral market in the future is very high. Various synthetic drugs and human-derived interferon are currently used as antiviral drugs, but they have many limitations in clinical practice. Synthetic drugs have a problem of increasing tolerability when used continuously, and interferon is used for various types of viruses, but its effectiveness is quite limited, and several side effects have been reported. Most of us experienced the disease progresses due to excessive inflammation such as a cytokine storm in the COVID-19 pandemic, ImmuneMed expects to be able to pioneer a new antiviral drug market with the commercialization of burfiralimab, anti-viral and anti-inflammatory therapeutics which will overcome the shortcomings of existing antiviral drugs.